COVID-19 vaccines: Global collaborations & concerted scientific effort sets record benchmark for pace of development
The COVID-19 pandemic and consequent economic and public health impact ravaged most of the year gone by. The silver lining is the spirit of collaboration and the power of science that has underlined the global quest for a solution. Pace of development of the COVID-19 vaccine will go down in history as a new benchmark.
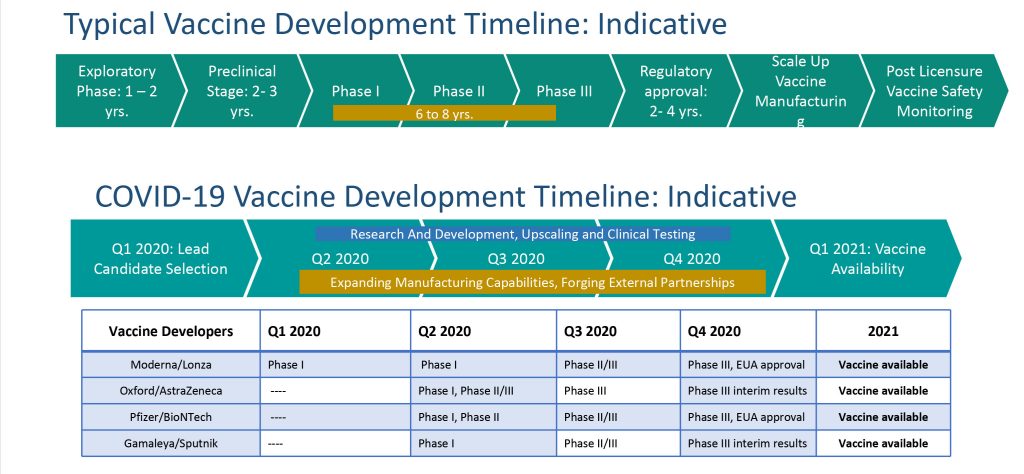
By March 2020, the month when WHO declared COVID-19 as a pandemic, 30 vaccine candidates were already under development globally and two (Moderna and CanSino) had already commenced Phase I clinical trials (Sathguru’s March 2020 roundup). At every milestone in the vaccine development journey new benchmarks have been set; and resolute efforts from early 2020 were critical for scripting the current record. This gets emphatic when seen against the context of SARS epidemic in 2003 when it took almost three months just to get the viral genome sequenced.
We now stand on a formidable pipeline of 232 vaccines with 60 in clinical development. 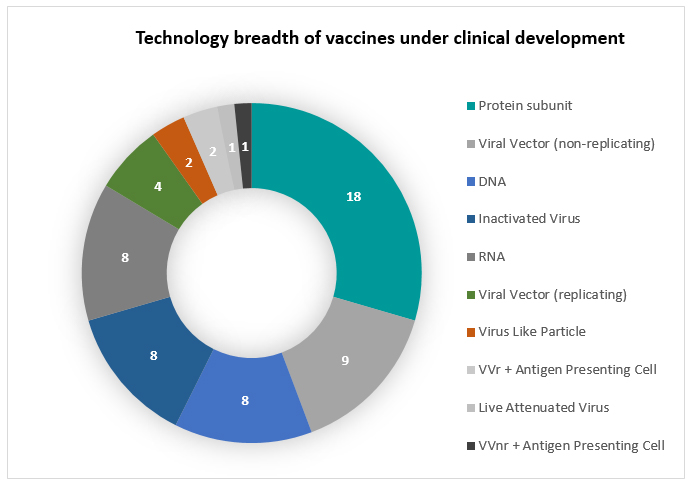 Creating history, several countries across the world have commenced vaccination for COVID-19 within nine months of it being declared a pandemic. The USFDA has approved Moderna and Pfizer-BioNTech vaccines, MHRA has approved Pfizer-BioNTech and AstraZeneca-Oxford vaccines, Russia has approved the Sputnik V, EMA has approved the Pfizer-BioNTech vaccine and China added to the list after approving Sinopharm’s vaccine couple of days before the year end. While global access remains a puzzle not fully addressed, the geographic spread of the Phase III pipeline is a great source of hope. We step into 2021 with great possibility of delivering to the world about 10 billion doses of the COVID vaccine from companies across geographic borders.
Creating history, several countries across the world have commenced vaccination for COVID-19 within nine months of it being declared a pandemic. The USFDA has approved Moderna and Pfizer-BioNTech vaccines, MHRA has approved Pfizer-BioNTech and AstraZeneca-Oxford vaccines, Russia has approved the Sputnik V, EMA has approved the Pfizer-BioNTech vaccine and China added to the list after approving Sinopharm’s vaccine couple of days before the year end. While global access remains a puzzle not fully addressed, the geographic spread of the Phase III pipeline is a great source of hope. We step into 2021 with great possibility of delivering to the world about 10 billion doses of the COVID vaccine from companies across geographic borders.
The journey has not been bereft of its fair share of setbacks and challenges. It was also a reminder of the reality of technology risk that is inherent in biopharmaceutical product development and translational research. Delays set back two fore-runners Inovio and Novavax that could have otherwise crossed the Phase 3 line by end of 2020. Clinical data disappointments impacted the Sanofi-GSK adjuvaneted vaccine, the University of Queensland-CSL program was halted due to potential interference with HIV screening and about a dozen other undeclared setbacks are folded into the preclinical long tail. And a big unknown looms on the world as 2021 dawns, the potential for efficacy against the mutated strain that emerged in UK and South Africa but now reported in several other countries including US and India.
Despite the setbacks, the global quest for COVID-19 vaccine is historic and will be remembered as a major scientific triumph. It has been no mean task to recalibrate the historical 8 to 10 year journey to under a year. And that too, while nurturing the technology breadth embodied in the global pipeline and ushering in the first ever mRNA vaccines (and potentially) the first DNA vaccines to get approved.
 Grow Beyond
Grow Beyond 

