Healthcare Newsletter – March 2020
We breathe life sciences and, we are data driven. We have been very disturbed by the global ‘infodemic’ and the barrage of rumours. The COVID – 19 pandemic is not an easy battle to win. The last thing we need is this noise around us that clouds the real facts. Instead of our monthly newsletter, we have summarized for you validated facts and details on COVID – 19 ’s current incidence and status of scientific response in the form of diagnostics, drugs and vaccines around the world.
With optimism that this will be behind us soon.
Team Sathguru
Source: WHO Coronavirus disease (COVID-19) Situation Dashboard
The COVID – 19 virus has been relentless and the global surge is uncontained at this point. The arsenal of scientific response will be determinant to how we tide through this wave of crisis. We discuss below evolving global landscape on development and adoption of diagnostics, drugs and vaccines for COVID – 19 . We have also included a spotlight on funding opportunities that can help us pool global resources and accelerate this journey to more sustainable victory.
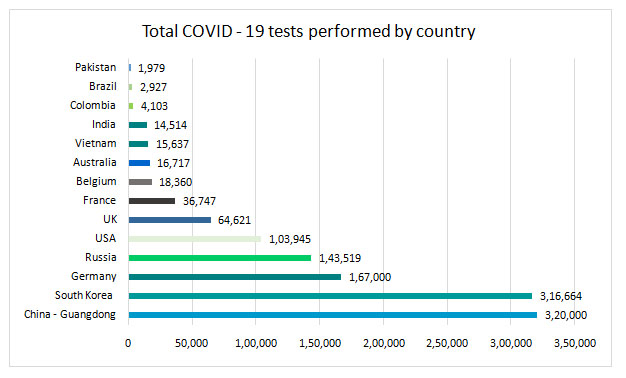
Testing and isolation will be cornerstones of response measures in most countries. Hence, access to diagnostic tests is a critical focus element for all public health authorities and governments. Diagnostic options available are an emphatic reflection of scientific advances achieved during the last decade. Within weeks of the virus first emerging, there has been access to molecular diagnostic tests both in the form of RT- PCR as well as whole genome sequencing. There is high level of promise of specificity and sensitivity. Serology tests, where conducted, have only been for preliminary assessment and have not been treated as confirmatory in any country. This is true even of testing in low and lower middle income countries (LMIC) where quick turn around of molecular tests has been a reality. India, for instance has adopted PCR testing in all regional pathology laboratories and whole genome sequencing (WGS) in national labs.
However, up until now, test availability has been a cause for concern globally. Global supply is still ramping up as several countries and regulators are now approving locally developed PCR test kits. With this swiftness of response from regulatory authorities all around the world and globally expanding supply, we stay optimistic that diagnostic preparedness over the next few weeks is likely to lesser of a concern.
United States of America: The USFDA declared Emergency Use Authorization (EUA) of in vitro diagnostic kits for detection/ diagnosis of the virus that causes COVID – 19 on February 4th and guidance documents released on February 29th. As of now, 16 companies have received authorization letters to manufacture and supply diagnostic test kits. However, due to controversy around access constraints for diagnostics, FDA has additionally permitted laboratory developed tests (LDT’s) and companies that are yet to obtain EUA to also serve the current need. Various healthcare systems and academic laboratories are now offering laboratory developed tests (LDT’s) and prescription decision rests with the clinician. In several states, this has helped substantially improve access to diagnostics, reliability albeit could be an open question.
India: With the growing number of cases in the country, the Indian Council of Medical Research (ICMR) quickly expanded testing strategy to include private labs with NABL accreditation to run real time PCR’s for the detection of the virus causing COVID – 19. The national task force has also capped the cost for testing while encouraging private players to engage in subsidized prices during this time of public health emergency. While PCR kits used until now were imported, in addition to allowing sale of any US FDA or European CE marked kit, ICMR designated institutions have now validated 14 kits and intimated the regulator CDSCO for approval.
Philippines: The Philippines FDA took active measure to ensure supply of a validated diagnostic test kits to detect the SARS – CoV – 2 virus by approving the use of test kits developed locally. The PCR detection kits were developed at University of the Philippines National Institute of Health (UP – NIS), intended for field testing.
Iran: Similar measure were taken in Iran with the Pasteur Institute of of Iran validating the SARS – CoV – 2 diagnostic testing kits developed by the Iranian Defense Ministry’s medical experts and engineers. The tests are set to be mass produced and distributed throughout the country.
Europe: European regulators have been actively approving foreign test kits since the beginning of this epidemic. Multiple COVID – 19 test kits have been reviewed and met with the CE marking in an accelerated approval process. The European Commission has also released multiple recommendations for testing strategies including suggesting that there could be merit in considering drive-in clinics for testing.

Emergence of rapid and point of care molecular tests: While we are encouraged by the widening availability of PCR test kits across the world, we are even more excited about emergence of sensitive molecular tests that can be done at the point of care. Rapid testing outside the hospital setup can reshape some of the critical response measures and create possibility for any community interventions. The most advanced of these is the ‘Cepheid Xpert Xpress SARS-CoV-2 test’. Developed by Danaher’s Cepheid, it received an emergency use authorization by the FDA on March 21st and is expected to be commercially available by March 30th. An accurate molecular test such as this which can be administered at the doorstep at the suspected person and provide results within an hour can be transformative and will alleviate pressure from pathology labs and other healthcare facilities. Mesa biotech followed in the same footsteps and received approval for similar POC kits on March 24th.
Testing continues to be a debated topic within response measures globally. Use of less reliable tests could cause more harm than good. False negatives can increase the risk of spread multifold and false positives would unnecessarily drain our healthcare capacity. Despite controversies around this topic globally, we are glad access to diagnostics is now well addressed and hope that high scientific threshold can continue to be maintained as long as this war against COVID – 19 is raged.

Since the initial detection in December 2019 in Wuhan, China, the new strain of Coronavirus (SARS – CoV – 2) causing COVID – 19 disease has spread rapidly across the globe. Given the heavy toll of the pandemic on the world, laboratories, researchers and pharmaceutical companies have actively been testing various new and repurposed drugs for disease treatment. Drugs previously approved for other viral infections that display potential for inhibitory effects on COVID – 19 virus are being tested in animal models and patients currently for effectiveness and safety. Repositioning an approved drug for this indication is a quicker process with greater probability for a near term solution that can be integrated in ongoing pandemic response measures. It is an attractive strategy due to the availability of existing supply chains for formulation manufacturing and distribution, facilitating lower costs and quicker commercial availability.
The ideal candidates so far have been anti-viral medications previously used for the treatment of SARS, MERS, and HIV due to the similarity of genomic layout and general replication kinetics of these viruses to the SARS – CoV – 2 virus. Other anti – malarial agents, interferon’s and anti-biotics have also been tested with some positive results.
While none of these are approved yet for therapeutic or prophylactic use for COVID – 19, the below table summarizes options currently under various stages of exploratory validation:
| Intervention | Previously Approved Treatment | Clinical Trials | Status |
| Chloroquine/ Hydrochloroquine | Malaria, Lupus, Rheumatoid Arthritis | Currently, chloroquine and hydroxychloroquine will be tested in patients with pneumonia | Preclinical studies found that the drug disrupts the ability of Covid – 19 virus to enter and replicate in human cells. Recent trial indicated 100% of patients treated with hydroxychloroquine in combination with the macrolide antibiotic azithromycin were virologically cured. |
| Combination: lopinavir/ ritonavir | HIV | Several trials involve combination treatment in comparison with the use of other drugs for COVID-19: Umifenovir, TCM, xiyanping, danoprevir-ritonavir and interferon inhalation | Improved clinical parameters in marmosets and mice infected with MERS-CoV but clinical studies displayed no statistical significance in drug group. |
| Umifenovir | Influenza Virus infections | Four clinical trials will be conducted for COVID-19 treatment: one in comparison with the basic treatment, and the other three studies comparing effects with oseltamivir, lopinavirritonavir, and carrimycin. | The Umifenovir anti-viral mechanism against influenza A and B involves viral fusion inhibition with the targeted membrane, which blocks virus entry into the cell. |
| Oseltamivir | Influenza Virus Infections | Undergoing a clinical trial with multiple combinations with chloroquine, ritonavir and favipiravir and a well-known as a broadspectrum antiviral drug. | Drug inhibits the viral neuraminidase and, consequently, blocks the release of viral particles from host cells, reducing the spread in the respiratory tract. |
| Remdesivir | Broad-spectrum antiviral treatment: nucleotide analog drug | Several phase 3 clinical trials are underway (NIH trial begins in Nebraska) for testing remdesivir in the United States, South Korea, and China. | It has been shown to inhibit replication of other human coronaviruses and efficacy in animal models has been demonstrated for SARS-CoV and MERS-CoV. |
| Interleukin-6 inhibitors | Rheumatoid arthritis | March 16: Sanofi and Regeneron announced initiation of a phase 2/3 trial of the IL-6 inhibitor sarilumab (Kevzara) among others. | An anti-interleukin-6 receptor monoclonal antibody (TZLS-501; Tiziana Life Sciences and Novimmune) is currently being developed. |
Apart from the mentioned interventions, other options have also been undergoing clinical trials in the fight against COVID – 19 Virus; human immunoglobulin, methylprednisolone, bevacizumab, thalidomide, vitamin C, pirfenidone, bromhexine, fingolimod, danoprevir, darunavir, cobicistat, and traditional Chinese medicines (TCM).
Given the expanding proportions of this public health emergency, interim results of these studies will yield important data that will help prioritize global efforts and accelerate approval of a solution. Global collaborations are more critical now than ever. This was evident when the European Medicines Agency (EMA) also emphasized urgent ‘‘need for large, multi-national trial protocols’’ and urged the medical research community to perform ‘‘large randomised controlled clinical studies as these are most likely to generate the conclusive evidence needed to enable rapid development and approval of potential COVID – 19 treatments.’’ As we stand in hope, we look forward to global industry and research community uniting this effort to accelerate therapeutic possibilities to clinical milestones.

While anti-viral drugs and anti-malarial drugs are being clinically investigated to be repurposed for accelerated approvals and therapeutic intervention in coronavirus disease and are at a more advanced stage of development, momentum is strengthening around next-generation therapies for COVID – 19 . These include cell therapy and immunotherapy to treat severe complications. Reported in about 14 – 20% of the patients, severe complications are primarily due to immune-mediated inflammatory response leading to Acute Respiratory Distress Syndrome (ARDS) in cases of prolonged infection. Mesenchymal Stem Cell (MSCs) have immunomodulatory properties and counteract the inflammatory processes by downregulating the production of inflammatory cytokines. Australia based Mesoblast was the first to announce its plans to evaluate its off-the-shelf allogenic MSC candidate remestemcel-L. The therapy has previously been evaluated in Chronic Obstructive Pulmonary Disorder (COPD) in a 60-patient trial where it was well-tolerated and significantly reduced inflammatory biomarkers. Another study published in China, reported that 7 critically ill pneumonia patients were successfully treated and their functional outcomes improved on treatment with MSCs. The Israeli Ministry of Health approved per-patient compassionate use of placenta-based cell therapy product PLX-PAD by Pluristem, an Israeli regenerative medicine venture. Previous pre-clinical findings of PLX cells revealed therapeutic benefit in animal studies of pulmonary hypertension, lung fibrosis, acute kidney injury and gastrointestinal injury which are potential complications of the severe COVID – 19 infection.
AlloVir and Baylor College of Medicine are collectively working on the development of off-the-shelf virus targeting T-Cell therapies against the novel coronavirus specifically for use in immune-compromised patients. They aim to position the SARS-CoV-2-specific T-cells for use as a monotherapy and incorporate the coronaviruses into ALVR106, a preclinical asset aimed at eliminating four community-acquired respiratory viruses: respiratory syncytial virus, influenza, para-influenza and human meta-pneumovirus.
Apart from cell and immunotherapies, monoclonal antibody based therapies are also being explored. Junshi Biosciences and Institute of Microbiology of the Chinese Academy of Sciences have verified the virus blocking activity of neutralizing antibodies (NAbs) in preliminary in vitro and in vivo activity and are due to fill Investigational New Drug (IND) application with domestic and overseas regulatory bodies. The advantage of NAbs is that they can used both as a prophylactic and therapeutic intervention and can be mass produced, making them suitable for use as a pandemic response measure.
Antibody based immunosuppressive drugs are also being explored to modulate the aggressive immune response leading to severe lung damage. US FDA gave Roche the green signal to conduct phase 3 Trial for its drug Actemra (Tocolizumab) which is approved for use in rheumatoid arthritis. Actemra is an interleukin 6 inhibitor that’s being repurposed to treat patients with severe lung damage due to ARDS. Sanofi and Regeneron have also kicked off a global testing program for Kevzara, their rheumatoid arthritis drug.
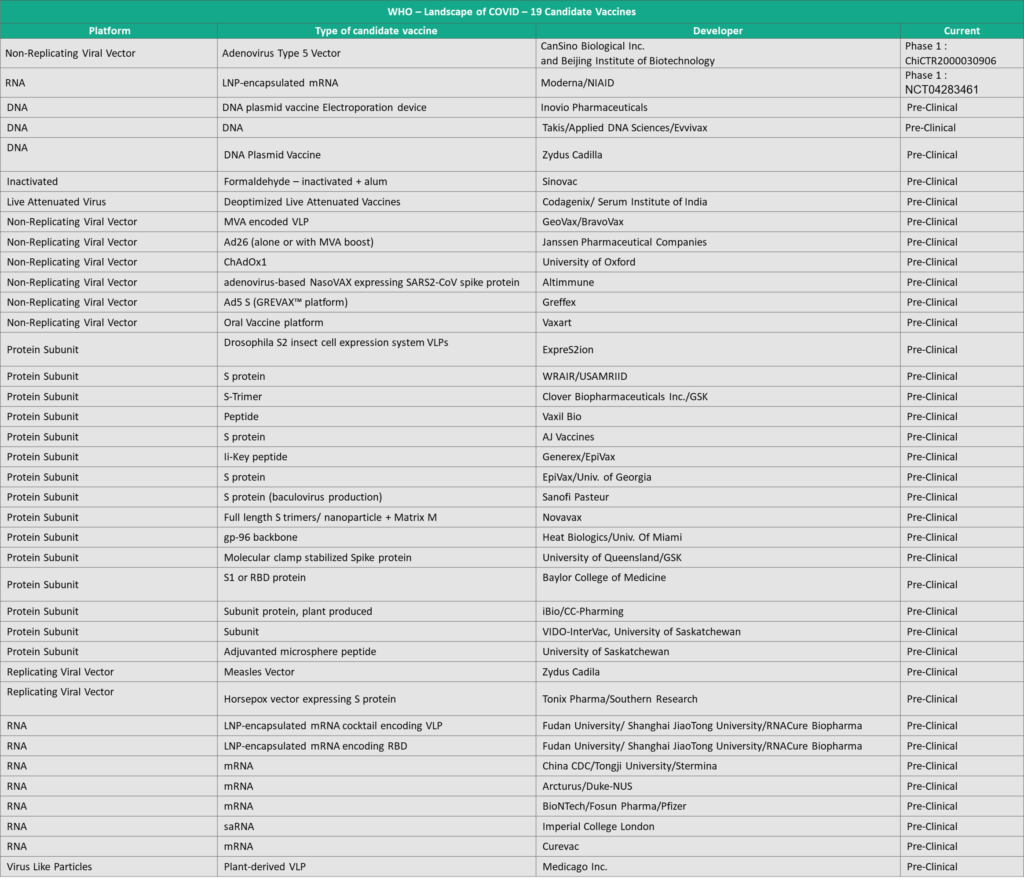
In the weeks leading to Coronavirus transforming into a global health crisis, clinicians, researchers and the biopharma companies have been scrambling to get a cure to the market. Scientific community around the world has touted the speed at which the whole genome sequence of the SARS-CoV-2 was made available to the world ‘unprecedented’ and can thus enable more accelerated response measures to this pandemic. By contrast, during the SARS Coronavirus outbreak in 2003, this took almost three months just to get the viral genome sequenced. We note with excitement that lead two lead vaccines in the world have already commenced clinical trials and the pipeline is robust.
Global pipeline:
Among a long list of companies currently developing a vaccine against COVID – 19, Moderna and CanSino Biologics are the front-runners with their vaccine candidates already into phase 1 clinical trials.
Moderna’s vaccine mRNA-1273 is an unconventional mRNA vaccine based on the genetic sequence of the spike protein – a major surface protein that the virus uses to bind to a human cell receptor and acts as the doorway into the human cell, infecting it while the virus replicates. The target was identified and a vaccine was underway just days after the whole genome was sequenced using Next Generation Sequencing and made publically available by the Chinese Academy of Medical Sciences, Institute of Pathogen Biology. Moderna, in partnership with the National Institute of Allergy and Infectious Diseases (NIAID) administered the vaccine to the first patient in the phase 1 trial. mRNA is an emerging platform and provides advantages of speed of development and production scalability as compared to a conventional vaccines. It doesn’t require production of large volumes of viruses, and thus also reduces the risks of contamination with live cells. CanSino’s candidate, Ad5-nCoV is a recombinant adenovirus based viral vector vaccine has been jointly developed by CanSino and the Institute of Biotechnology of the Academy of Military Medical Sciences. The vaccine is now entering phase 1 trials with 108 patients who are permanent residents of the city of Wuhan, the epicentre of the outbreak in China.
There are over 30 other candidates in the pre-clinical stage of development based on recombinant viral vectors, protein subunits, plant based virus-like particles (VLP) and live attenuated vaccines. Live attenuated vaccines, although highly effective, are often not preferable for people with compromised or weakened immune systems due to the underlying risk of developing an infection. Hence, it is yet to be validated if a live attenuated vaccine for COVID – 19 would be suitable considering that the patients may already have weakened immune systems from a long-lasting infection. Biotechnology companies with proprietary vaccine platforms are an important stakeholder in the development due to the ability of the novel platforms to deliver efficiently in an emergency outbreak response. BravoVax, CC-Pharming, Evvivax own proprietary vaccine production platforms and have entered into partnerships with GeoVaxiBio and Takis respectively to develop vaccine against SARS-CoV-2.
Global efforts to accelerate vaccine development – Swift regulatory action & cooperation
Gravity of the global crisis calls for unprecedented levels of cooperation amongst countries and regulators. In a swift response, a regulatory workshop for SARS-CoV-2 vaccine development was held on the 17th March under the umbrella of International Coalition of Medicines Regulatory Authorities (ICMRA), co-chaired by the WHO, US FDA and EMA and attended by delegates from 17 countries. With a tangible outcome to moot, we now have very specific guidance (released on 24th March) on accelerating vaccine candidates to Phase I /first-in-human clinical trails and data needed to get to this critical milestone. Encouraging provisions of this guidance include:
- Vaccine using a well-characterised platform technology that has previously been used in the manufacturing of a licensed vaccine, toxicology data and other clinical data from the previously approved vaccine can be submitted.
- The vaccine manufacturer should have adequate data to support why certain preclinical studies may be omitted if sufficient historical data is available for a closely related vaccine product
- Necessary to obtain data on immune response in animals against SARS-CoV-2 vaccine candidate
- Not necessary to demonstrate efficacy of the vaccine in animal models
The guideline reaffirms commitment of global regulators to accelerated vaccine development and also pave the way for a more collaborative global effort. They strike the right balance between accelerating potential vaccine candidates to commercial use milestones but while addressing critical concern of safety of human subjects enrolled in these studies. With global collaboration gaining such momentum, we are hopeful that sustainable solutions for population level prophylaxis will soon be a reality.
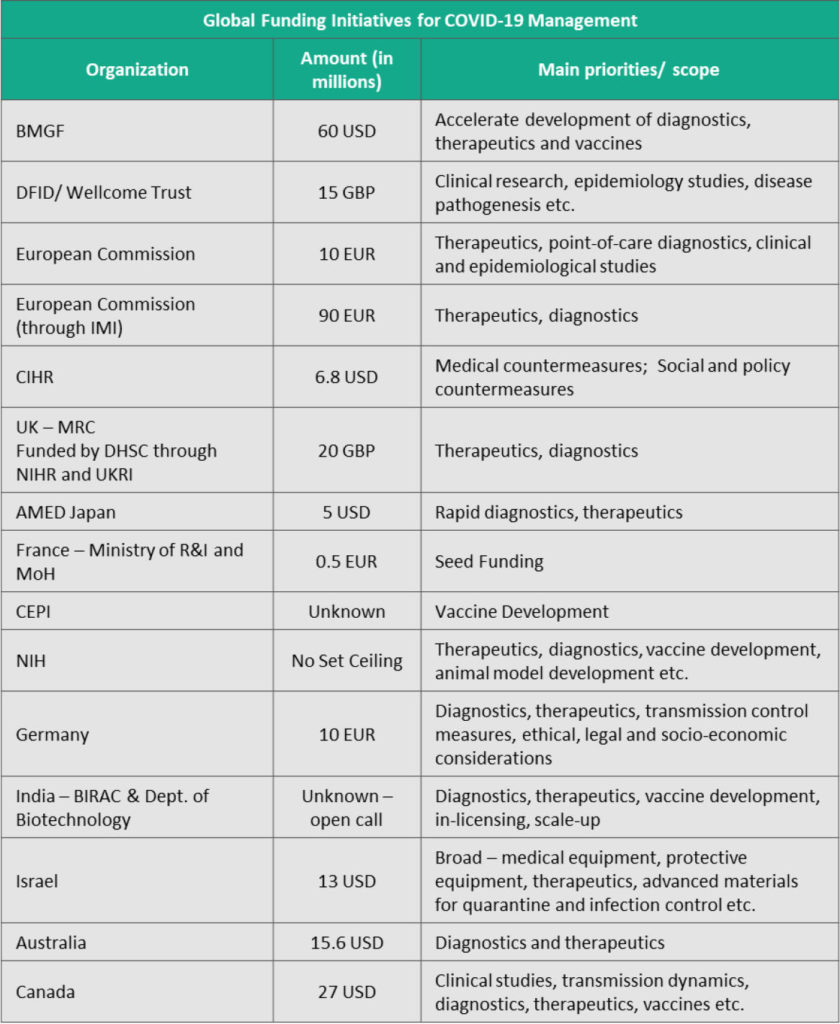
Governments and organizations across the world are coming together to strengthen global arsenal in this war against the Coronavirus. We have summarized below cross-stakeholder initiatives; and hope innovators across borders are able to pool resources and accelerate solutions to milestones where are can be globally implemented.
World Health Organization (WHO) has set up a COVID-19 Solidarity Respond Fund, which has currently raised more than $195 million. As a part of its Strategic Preparedness and Response Plan, among other activities, the donations will also go towards accelerating the development of vaccines, drugs and tests for coronavirus disease.
Bill and Melinda Gates Foundation, Wellcome Trust and Mastercard together launched the COVID – 19 Therapeutics Accelerator and committed to it up to $125 million in seed funding to accelerate the response to the pandemic by identifying, assessing, developing, and scaling-up treatments. The Accelerator will fund the innovations and development for drugs that can be developed, mass-produced and delivered rapidly. It will play a catalytic role by working with government sector funders, organizations as well as the regulatory and public health institutions to provide an end-to-end focus and support for drug pipeline development through manufacturing and scale-up. Early stage funding will de-risk the pathway for new and re-purposed drugs and biologics to make them readily available in low-resource settings for COVID – 19 and future epidemic threats as well. UK Government has set up a £20 million fund for research into Coronavirus disease and vaccine development. From the total sum, £2.2 million is being earmarked for a new recombinant adenovirus viral vector based vaccine development effort by the University of Oxford. The vaccine utilizes the same technique as a vaccine the team previously developed for the closely related MERS coronavirus, which showed promise in animal and early-stage human testing. Another £0.4 million is being used to fund the development of a manufacturing process of the same vaccine to produce it at a million-dose scale once the trials are successful.
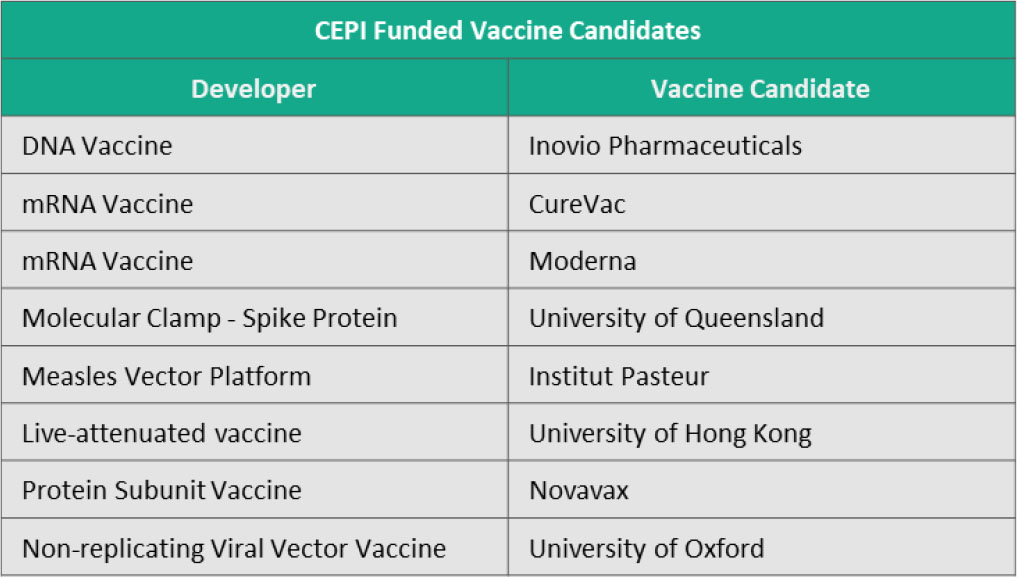
Coalition for Epidemic Preparedness Innovations (CEPI), a global partnership between public, private, philanthropic, and civil body organizations, is currently backing the development and clinical testing for eight vaccine candidates.
European Commission launched theSC1-PHE-CORONAVIRUS-2020: Advancing knowledge for the clinical and public health response to the [COVID-19] epidemic’ funding program on January 30th. 17 research projects involving 136 research teams from across EU have been shortlisted for the earmarked funding of €47.5 million. Projects shortlisted include solutions to improve epidemiology and public health, rapid point-of-care diagnostic tests, new treatments and new vaccines.
In India, Department of Biotechnology and Biotechnology Industry Research Assistance Council (BIRAC), are also inviting project proposals for developing diagnostics, vaccines, novel therapeutics, repurposed drugs etc. for controlling COVID – 19 extending the funding opportunity to internal development as well as in-licensing and scale-up projects.
Click here to Download the PDF version
Authors:



 Grow Beyond
Grow Beyond